Komei Washino, VMD, Ph.D. Chief Technology Officer, AMS Planning, Inc. Former Director, Nihon Medi-Physics | Research Professor, Hokkaido University Graduate School of Medicine | Senior Researcher, National Institutes for Quantum and Radiological Science and Technology (QST)
The Monitoring Gap in Radioligand Therapy
The approval of ¹⁷⁷Lu-PSMA-617 (Pluvicto) by the US Food and Drug Administration for metastatic castration-resistant prostate cancer (mCRPC) marked a significant milestone in radioligand therapy. Supported by the VISION and TheraP trials, and more recently by PSMAfore data supporting earlier-line use ,¹⁷⁷Lu-PSMA-617 has become an established option for patients who have progressed through androgen receptor pathway inhibitors and taxane-based chemotherapy.
Standard clinical practice relies on a baseline PSMA-PET/CT to confirm eligibility — assessing the adequacy and homogeneity of PSMA expression across disease sites before therapy begins. What happens between that baseline scan and the completion of subsequent treatment cycles has, until recently, received comparatively less systematic attention.
This is the gap that a study published in the Journal of Nuclear Medicine begins to address
The Study: Post-Therapy SPECT/CT as a Window into Interim Disease Change
Parihar and colleagues at Washington University School of Medicine retrospectively analyzed 79 men with mCRPC treated with ¹⁷⁷Lu-PSMA-617, each of whom had evaluable baseline PSMA-PET/CT and post-first-cycle SPECT/CT imaging available for comparison [1].
The central finding was striking in its frequency: 38% of patients showed discordant findings between their post-therapy SPECT/CT and their baseline PET/CT. These discordances included:
- New lesions exclusively: 8 patients
- Enlarging existing lesions only: 12 patients
- Both new and enlarging lesions: 10 patients
- New hydronephrosis requiring ureteric stenting: 2 patients
- New spinal cord compression requiring corticosteroids: 1 patient
The anatomic distribution of new or enlarging lesions spanned nodal (11), bone (20), and visceral (9) sites — reflecting the systemic nature of disease evolution in this population.
Critically, the likelihood of discordance was strongly associated with the time elapsed between baseline imaging and treatment. The median interval from baseline PET/CT to post-therapy SPECT/CT was 88 days among patients with discordant findings, compared to 40 days among those with concordant findings (p=0.001, Kruskal-Wallis test). Of all discordances identified, 93.3% occurred in patients whose first therapy cycle was performed more than 30 days after the baseline PET/CT.
The authors concluded that for patients with a baseline PSMA-PET/CT obtained more than 30 days before therapy, a post-first-cycle whole-body SPECT/CT should be performed — both as a new baseline for future comparisons and as a source of adjunct clinical information. They further suggested that the baseline PSMA-PET/CT should ideally be obtained within 4 weeks of the first therapy cycle.
What This Finding Means Pharmacokinetically
The 38% discordance rate is not simply a logistical inconvenience. It reflects something more fundamental: disease biology and drug distribution are dynamic, and a single pre-treatment snapshot — however sophisticated — cannot fully anticipate what will be observed once therapy is underway.
This has pharmacokinetic implications that extend beyond imaging. ¹⁷⁷Lu-PSMA-617, like other radioligand therapies, distributes across the body according to the interplay of tumor uptake, normal tissue binding, and renal excretion. The same factors that drive discordance in SPECT/CT findings — changes in tumor burden, receptor expression, and disease distribution — are also factors that influence how the drug behaves pharmacokinetically within each individual patient.
The Parihar study focuses, appropriately, on spatial distribution: where is the disease, and how has it changed? SPECT/CT is well-suited to answer this question. What it cannot directly answer is the complementary temporal and quantitative question: how is the drug being eliminated, and at what rate, and does that rate vary between patients in ways that correlate with treatment response or disease dynamics?
These are distinct but related questions. Spatial imaging and excretion dynamics are not competing approaches — they are different dimensions of the same pharmacokinetic reality.
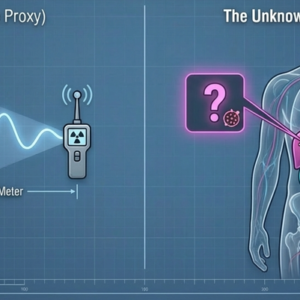
Urinary Excretion as a Complementary Research Dimension

In radioligand therapy with ¹⁷⁷Lu-based agents, urinary excretion represents the dominant elimination pathway. Academic research has begun to explore whether the timing and rate of urinary excretion might encode information about individual pharmacokinetic behavior — information that is, by definition, unavailable from the current clinical setting of spatial imaging alone.
If a patient shows discordant post-therapy SPECT/CT findings — new lesions, unexpected tumor growth, evidence of disease progression between cycles — one reasonable research question is whether any signal in the urinary excretion profile might have preceded or accompanied that change. Do patients with higher tumor burden eliminate the drug differently? Do patients who show early disease progression exhibit distinct excretion kinetics?
These questions are genuinely open. The literature does not yet provide clear answers, and the methodological work required to pursue them rigorously — standardized collection, validated measurement, appropriately controlled study designs — remains to be done.
What the Parihar study contributes is a compelling demonstration that individual variability in disease course is not only real but clinically consequential, occurring in more than one-third of patients within the first treatment cycle. That variability, observed spatially through SPECT/CT, raises natural questions about what other dimensions of individual pharmacokinetic behavior might be worth examining.
Those working in nuclear medicine facilities encounter post-therapy urine as a routine operational reality. The questions that the academic literature is beginning to ask about dynamic monitoring and individual pharmacokinetic variability are questions that arise, in a different register, from direct engagement with that material every day. The distance between operational observation and systematic research inquiry may be shorter than it appears.
A Field in Motion
The Parihar study is a meeting report based on retrospective, single-institution data. Its findings should be interpreted with appropriate caution regarding generalizability. What it offers is a well-characterized observation and a clear hypothesis: that the interval between baseline imaging and treatment delivery matters, that disease can change meaningfully within that interval, and that post-therapy imaging provides information that pre-therapy imaging cannot.
The broader implication is one that the field of radioligand therapy is increasingly recognizing: monitoring should be dynamic, not static. A single baseline assessment, however comprehensive, is a starting point rather than a complete picture.
What the full picture looks like — spatially, temporally, quantitatively — remains an open and active area of research. We find it worth following closely.
Reference [1] Parihar AS, Dehdashti F, Pachynski R, Picus J, Reimers M, Pant N, Gay H, Kim H, Rangbulla M, Jahromi A, Swingle C, Michalski J, Wahl R, Prasad V. Clinical utility of post-therapy SPECT/CT after the 1st cycle of ¹⁷⁷Lu-PSMA-617 therapy in detecting interim disease progression compared to pre-therapy PSMA PET/CT. J Nucl Med. 2025;66(suppl 1):251407. https://jnm.snmjournals.org/content/66/supplement_1/251407
This content is prepared for general academic discussion purposes only. It does not constitute medical advice, clinical guidance, or diagnostic recommendations. Treatment decisions must be made exclusively by licensed medical professionals. This content does not constitute promotion of any product or service. AMS Planning, Inc. makes no warranty regarding the completeness or accuracy of the information presented.
About the Author: Komei Washino, VMD, Ph.D., is Chief Technology Officer at AMS Planning, Inc. He brings 30 years of experience in the development, research, and commercialization of radiopharmaceuticals. Prior to joining AMS, he served as Director at Nihon Medi-Physics, held an appointment as Research Professor at Hokkaido University Graduate School of Medicine, and served as Senior Researcher at the National Institutes for Quantum and Radiological Science and Technology (QST). He received his VMD and Ph.D. from the Faculty of Veterinary Medicine, Hokkaido University.