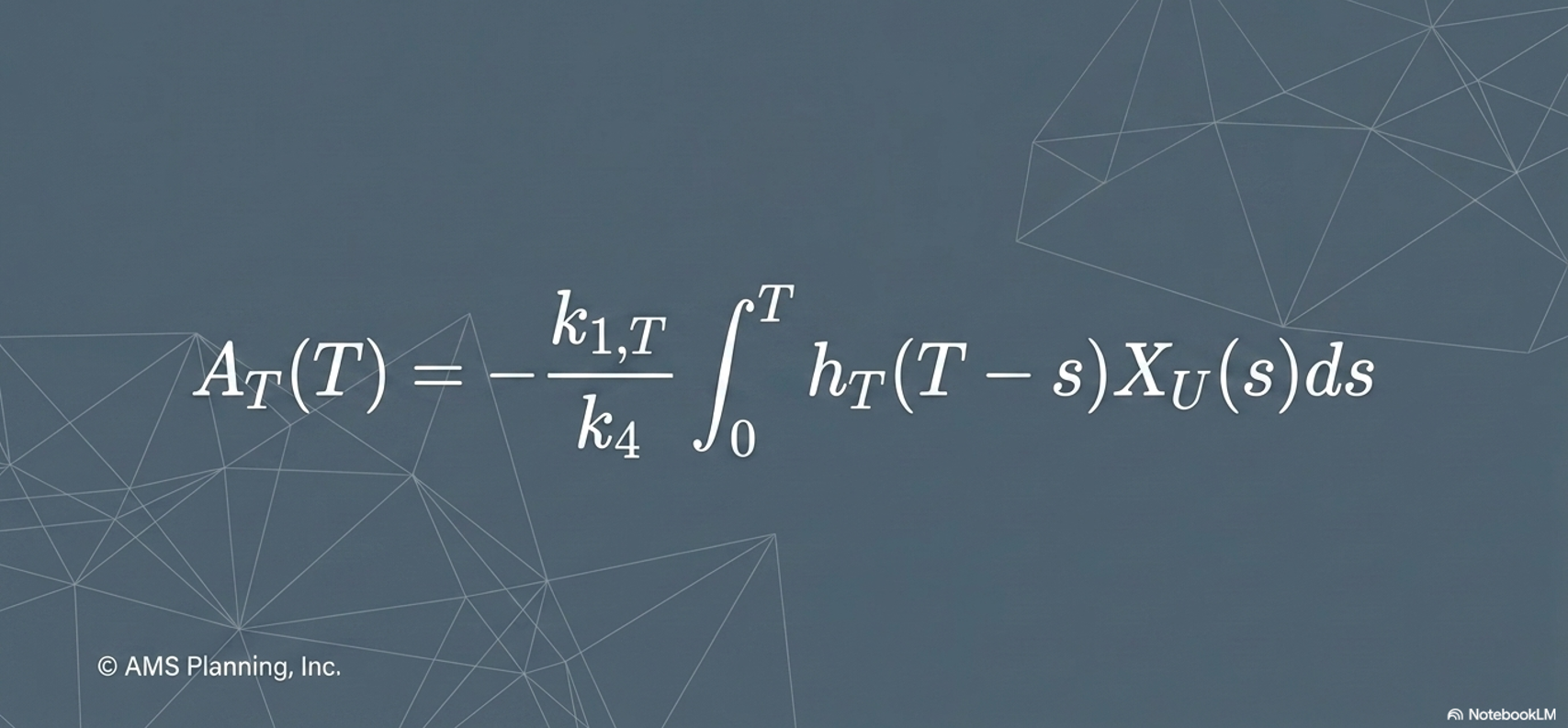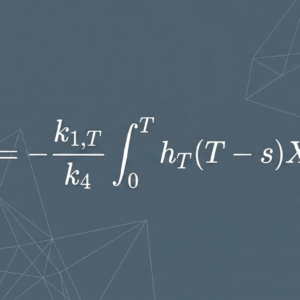Komei Washino, VMD, Ph.D. Chief Technology Officer, AMS Planning, Inc. Former Director, Nihon Medi-Physics | Research Professor, Hokkaido University Graduate School of Medicine | Senior Researcher, National Institutes for Quantum and Radiological Science and Technology (QST)
A Question Worth Asking
In conventional oncology, treatment response is typically assessed weeks after therapy begins — RECIST criteria, for instance, evaluate solid tumor response after 6 to 8 weeks of follow-up. For patients undergoing radiotherapy with ¹⁷⁷Lu-labeled agents, this means a significant period of uncertainty between administration and the first formal assessment of whether the therapy is working.
This raises a question that, to our knowledge, has received insufficient systematic attention:
Is there information available earlier — perhaps within the first 48 hours after administration — that could tell us something meaningful about how a given patient’s body is responding to treatment?
We believe there may be. And we believe the answer may be, at least in part, hiding in the urine.
The Pharmacokinetic Logic
The therapeutic efficacy and toxicity of ¹⁷⁷Lu-labeled radiotherapy agents arise not from the pharmacological properties of the carrier molecule, but from the biological effects of radiation emitted by the radioisotope retained within target tissues. The critical variables are: how much of the agent reaches the target, how long it stays there, and how much is eliminated before it can exert its intended effect.
For agents like ¹⁷⁷Lu-DOTATATE and ¹⁷⁷Lu-PSMA-617, the primary elimination pathway is renal. Quantitative data from prescribing information and pharmacokinetic studies indicate that a substantial fraction of administered radioactivity is excreted in urine, predominantly within the first 24 hours post-administration.
This creates a fundamental pharmacokinetic relationship: the rate and extent of urinary excretion could be, in principle, inversely related to the amount of drug distributed to and retained in target and non-target tissues.
In principle, faster excretion would be associated with a smaller area under the curve (AUC) in blood target organs, thus, a lower radiation dose — and potentially, though not necessarily, with attenuated efficacy. The reverse would be expected with slower excretion, which could be associated with a larger AUC and higher radiation dose, but also with greater exposure to non-target organs and associated toxicity risk. Whether this theoretical relationship holds in clinical practice, and to what degree, remains to be established.
If this relationship holds under defined conditions, cumulative urinary radioactivity excretion may serve as a research variable reflecting individual pharmacokinetic behavior — one that could, if validated, contribute to our understanding of the factors influencing efficacy and toxicity.
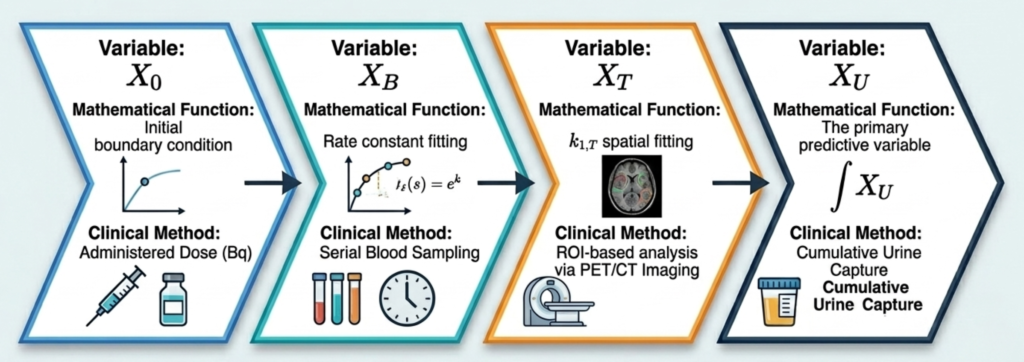
A Five-Compartment Model: Formalizing the Relationship
To explore this hypothesis rigorously, we developed a five-compartment pharmacokinetic model designed to describe the distribution and urinary excretion of radiotherapy agents labeled with alpha or beta-emitting radionuclides in humans.
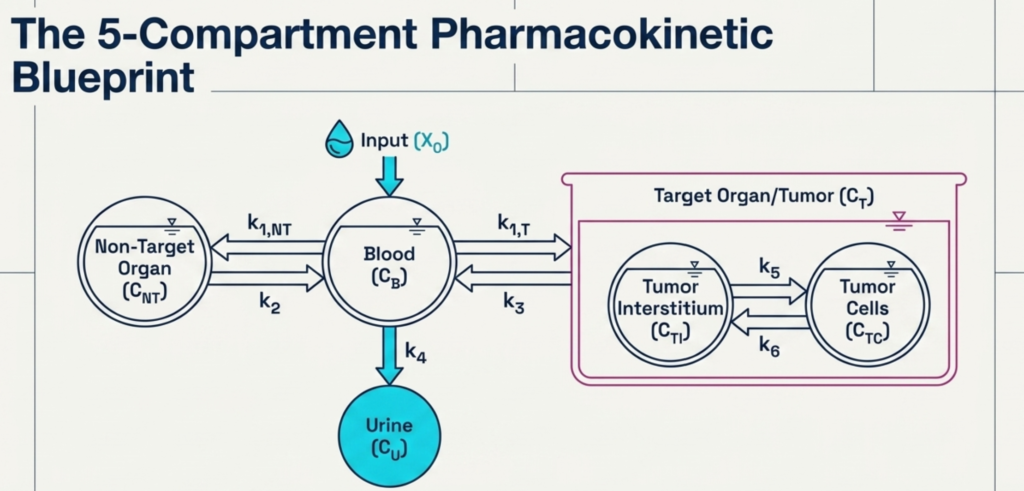
The model incorporates the following compartments:
- Blood (C_B): whole blood or plasma
- Non-target organ(s) (C_NT): peripheral tissue(s) relevant to dose-limiting toxicity — such as bone marrow, salivary glands, intestinal tract, etc.
- Target organ (C_T): subdivided into tumor interstitium (C_TI) and tumor cells (C_TC), depending on the binding characteristics of the carrier molecule.
- Urine (C_U): treated as cumulative excretion
The model is governed by differential equations describing transfer rate constants (k) between compartments, capturing the dynamics of vascular distribution, tissue uptake, cellular internalization, and renal elimination.
Four assumptions underlie the model: single intravenous bolus administration; absence of prolonged non-specific retention; negligible secondary metabolite distribution; and renal-urinary as the primary elimination route. These are consistent with the known pharmacokinetic behavior of ¹⁷⁷Lu-labeled agents at the nanogram-level chemical doses used clinically — well below the saturation threshold of target molecules, supporting the assumption of linear pharmacokinetics.
The Mathematical Relationship
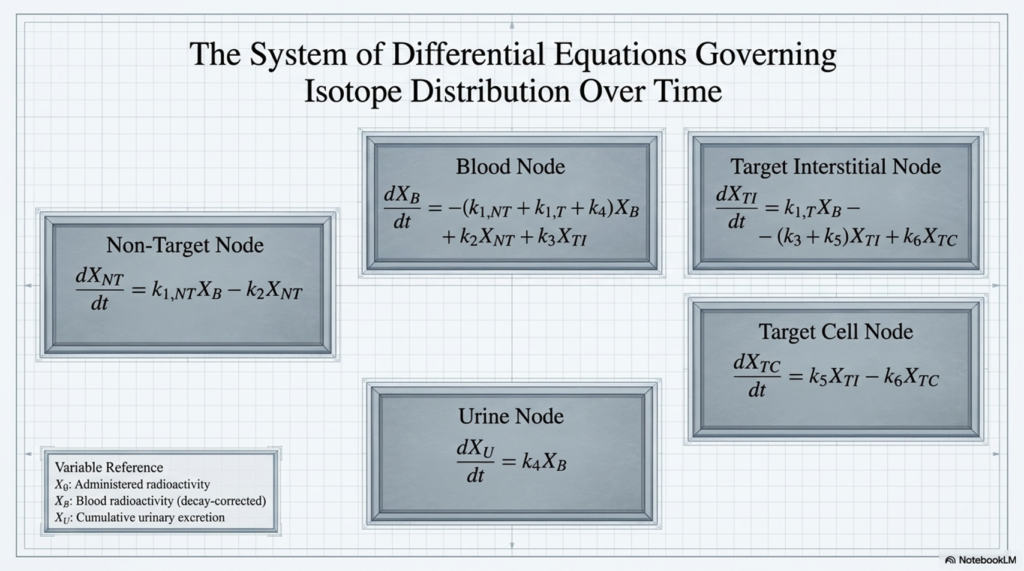
The central insight of the model is this: the amount of drug present in the target compartment at any time t, and the AUC of the target compartment over any defined time window, can be expressed mathematically as a function of the cumulative urinary RI excretion up to that time. The same relationship holds for the non-target compartment.
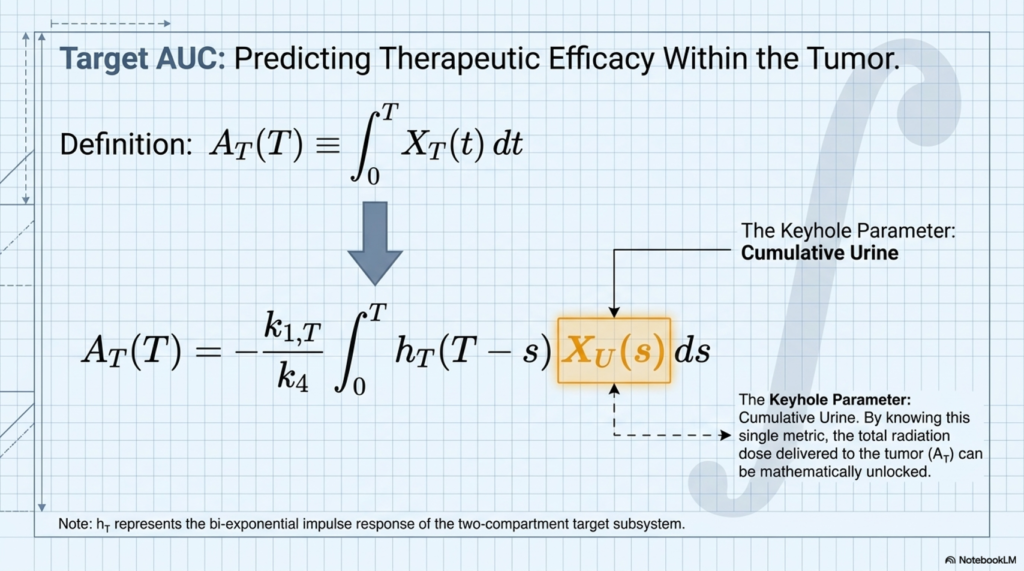
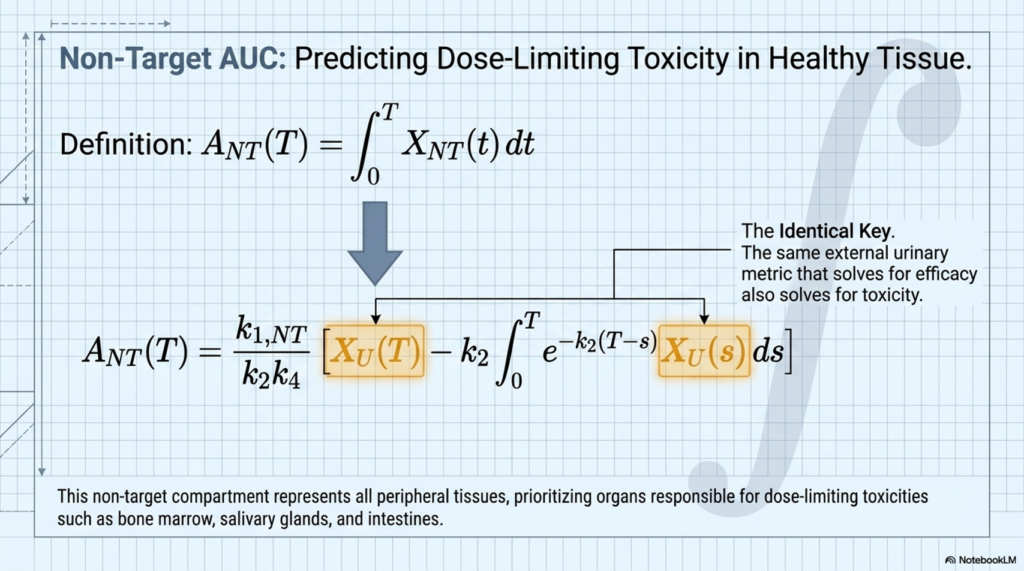
This means that a sufficiently precise measurement of cumulative urinary radioactivity excretion — particularly over the first 48 hours post-administration — could serve as a quantitative proxy for AUC in both tumor and toxicity-relevant organs.
The 48-hour window is not arbitrary. The effective half-life (T_eff), integrating both physical decay and biological clearance, determines the period during which a radiotherapy agent can deliver a clinically meaningful radiation dose. For medium-molecular-weight ¹⁷⁷Lu-labeled agents, this window encompasses approximately three to four effective half-lives — spanning from immediately after administration to approximately 48 hours.
What This Could Mean in Practice
If the inverse correlation between cumulative urinary excretion and organ AUC is empirically confirmed through prospective clinical study, several implications follow. Each represents an area for future research rather than established clinical practice.
Early pharmacokinetic phenotyping. Measuring cumulative urinary radioactivity over the first 24–48 hours after the first treatment cycle could, in principle, stratify patients by individual pharmacokinetic profile before the second cycle.
Modulation of excretion kinetics. The pharmacokinetics of radiopharmaceuticals are known to vary with hydration status and concomitant medications. If the urinary excretion rate constant (k₄) can be modulated by controlling water intake timing and volume after administration, this could theoretically influence the balance between tumor retention and excretion — a hypothesis warranting prospective investigation.
Earlier response signal. Rather than waiting 6–8 weeks for imaging-based response assessment, urinary excretion data collected within 48 hours might provide an earlier pharmacokinetic signal. Whether this signal correlates with eventual treatment response is an open question the model frames, but does not answer.
These are theoretical implications of a mathematical model. Experimental verification through prospective clinical studies is required.
Why This Question Has Been Hard to Ask
There is a practical reason why cumulative urinary radioactivity has not been systematically studied as a pharmacokinetic variable in ¹⁷⁷Lu therapy: measuring it is operationally challenging.
Post-therapy urine is classified as radioactive waste. Standard practice involves storage and decay. Precise quantitative measurement of radioactivity across defined collection intervals — the kind of data required to construct a meaningful cumulative excretion curve — is not part of routine clinical workflow.
Advances in radioactive wastewater management technology are beginning to change this operational reality. Systems capable of capturing and quantifying urinary radioactivity within defined time windows are now technically feasible. Whether the data generated through such systems could, with appropriate study design, contribute to the pharmacokinetic research questions framed here is a question we find genuinely interesting — and intend to pursue.
Those working in nuclear medicine facilities encounter post-therapy urine as a daily operational reality. The pharmacokinetic questions raised here are not abstract — they arise directly from that practical context.
An Invitation to the Field
The model presented here is a theoretical framework. It is built on established pharmacokinetic principles and supported by the known biology of ¹⁷⁷Lu-labeled agents. It has not yet been validated against clinical data.
What it offers is a formalization of a question the field has not yet asked with sufficient rigor: what does post-therapy urine tell us about what is happening inside the patient?
We believe this question is worth asking. We believe the infrastructure to answer it is becoming available. And we believe the answer — whatever it turns out to be — will matter for the future of precision radiopharmaceutical therapy.
Future prospective clinical studies examining the correlation between cumulative urinary RI excretion and treatment response are, in our assessment, warranted.
Note on methodology: The pharmacokinetic model described in this article represents a theoretical framework developed by AMS Planning, Inc. It was not derived from specific experimental results. The scientific validity of the model will be evaluated through future experimental verification. This content is prepared for general academic and scientific discussion purposes only. It does not constitute medical advice, clinical guidance, or diagnostic recommendations. Treatment decisions must be made exclusively by licensed medical professionals. This content does not constitute promotion of any product or service. AMS Planning, Inc. makes no warranty regarding the completeness or accuracy of the information presented.
About the Author Komei Washino, VMD, Ph.D., is Chief Technology Officer at AMS Planning, Inc. He brings 30 years of experience in the development, research, and commercialization of radiopharmaceuticals. Prior to joining AMS, he served as Director at Nihon Medi-Physics, held an appointment as Research Professor at Hokkaido University Graduate School of Medicine, and served as Senior Researcher at the National Institutes for Quantum and Radiological Science and Technology (QST). He received his VMD and Ph.D. from the Faculty of Veterinary Medicine, Hokkaido University.