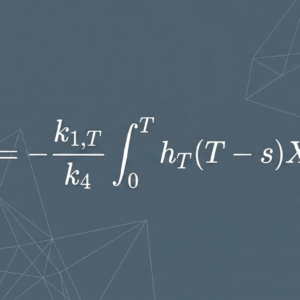Komei Washino, VMD, Ph.D. Chief Technology Officer, AMS Planning, Inc. Former Director, Nihon Medi-Physics | Research Professor, Hokkaido University Graduate School of Medicine | Senior Researcher, National Institutes for Quantum and Radiological Science and Technology (QST)
The Logistical Reality of PRRT
Peptide receptor radionuclide therapy (PRRT) with ¹⁷⁷Lu-Dotatate has become a cornerstone treatment for neuroendocrine neoplasms. Its clinical efficacy is well-established. Yet a practical challenge remains at the center of its wider adoption: radiation safety management after administration.
In Japan, patients must remain in dedicated isolation facilities until their surface dose rate falls below 18 μSv/h before discharge is permitted. This requirement places real constraints on hospital capacity and, more importantly, on patient access to therapy. As of 2022, Japan had only 66 dedicated nuclear medicine rooms and 160 beds to serve this need — a resource gap that is expected to widen given the expanding evidence base for ¹⁷⁷Lu-Dotatate, including recent data from the NETTER-2 trial supporting its use in first-line settings.
The question of how to predict — and eventually optimize — this waiting period has become an active area of clinical and pharmacokinetic research.
A Practical Predictive Model: The Yamagata University Study
A recent prospective study from Yamagata University offers a compelling contribution to this challenge [1]. Researchers investigated which variables could reliably predict the decline in surface dose rate at 5.5 hours post-administration of ¹⁷⁷Lu-Dotatate, studying 13 patients who received a standardized dose of 7.4 GBq.
Their findings were notably suggestive. Among the variables examined, just two emerged as significant predictors: maximum tumor diameter and renal function, measured by creatinine clearance using the Cockcroft-Gault equation.
The resulting prediction formula is explicit:
Dose rate ratio (%) = 51.4 + 0.360 × maximum tumor diameter (mm) − 0.212 × creatinine clearance (ml/min)
This formula demonstrated an extremely strong correlation with observed outcomes (R² = 0.937, p < 0.001), with a prediction error of only −5.5% to 4.2% across the study cohort.
The pharmacokinetic logic is straightforward. ¹⁷⁷Lu-Dotatate binds to somatostatin receptors expressed on neuroendocrine tumors. Larger tumors — with greater receptor-expressing volume — retain more radioactivity for longer periods, delaying the decline in surface dose rate. Simultaneously, the kidneys serve as the primary excretion pathway; patients with higher creatinine clearance eliminate the drug more rapidly. Together, these two variables capture the essential tension between retention and elimination.
The practical implication is significant. A clinician with knowledge of a patient’s tumor diameter and renal function can, in principle, estimate discharge timing before therapy even begins. The authors further demonstrated that one patient in the cohort — with a tumor diameter of 29mm and a creatinine clearance of 104.2 ml/min — met the discharge threshold within 5.5 hours and subsequently received their third and fourth treatment cycles on an outpatient basis.
What the Model Reveals — and What It Cannot
The elegance of this predictive model invites a deeper question. If tumor diameter and renal function explain 93.7% of the variance in dose rate decline (R² = 0.937), what accounts for the remainder?
The academic literature on radiopharmaceutical pharmacokinetics consistently documents inter-individual variability that extends beyond any single predictive model. The Yamagata study itself notes that variations in water intake, tumor microvasculature, receptor expression heterogeneity, and differences in tissue distribution may all contribute to outcomes that the formula cannot fully anticipate.
Surface dose rate, for all its practical utility, is an external measure. It reflects the aggregate of what is happening inside the body — retention in tumor tissue, distribution across healthy organs, and excretion through the kidneys and bladder — without directly distinguishing between them. Two patients with identical surface dose rates at 5.5 hours may have arrived at that measurement through quite different internal kinetic pathways.
The authors themselves acknowledge this: they describe their study as preliminary, conducted at a single institution with a small and heterogeneous sample, and call for future analysis using larger patient numbers and multiple SPECT/CT measurements. This is not a limitation unique to this study — it reflects the current state of the field.
The Role of Urinary Excretion Data in Pharmacokinetic Research
This is where urinary excretion data enters the academic conversation.
Urine has long served as a research matrix in pharmacokinetic and toxicology studies across therapeutic areas. In the context of ¹⁷⁷Lu-Dotatate, the kidneys are the dominant excretion pathway — a fact central to the Yamagata model itself. Yet the study, by design, measured renal function indirectly through creatinine clearance rather than through direct observation of urinary excretion dynamics during and after administration.
Academic research has begun to explore whether patterns in urinary excretion — the timing, the rate, the cumulative amount excreted across defined intervals — might serve as a more direct window into individual pharmacokinetic behavior. If a patient’s surface dose rate is declining more slowly than the formula predicts, is that because tumor retention is higher than anticipated? Or because urinary excretion is delayed? These two scenarios have different implications, yet they may present identically on surface dosimetry.
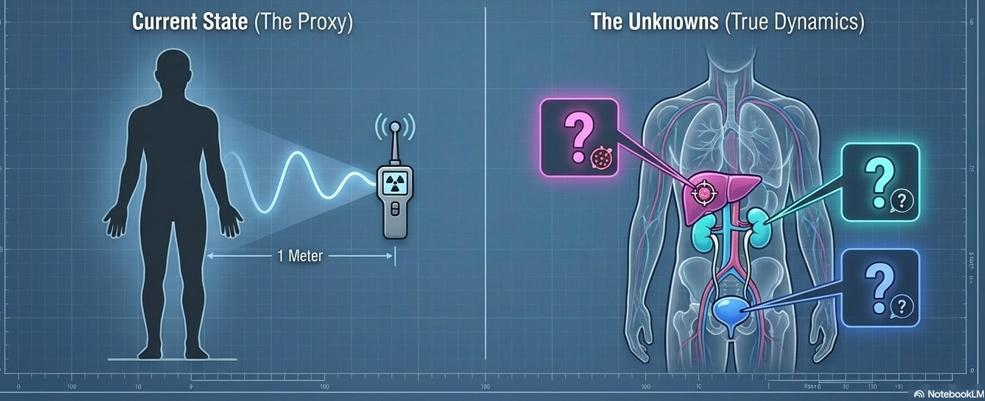
Urinary excretion data, analyzed in conjunction with predictive models such as the one developed at Yamagata University, could in principle help researchers begin to disentangle these contributions. Understanding not just how much radioactivity remains, but when and how it is being eliminated, represents a natural extension of the pharmacokinetic inquiry this study opens.
The methodological challenges are real: standardizing collection protocols, accounting for hydration variability — which the Yamagata authors themselves flag as a relevant factor — and developing validated analytical frameworks are all prerequisites for meaningful research in this space.
Those working in nuclear medicine facilities encounter these questions in practical, operational contexts every day. The gap between the academic discussion and the empirical reality on the ground is, in our observation, smaller than the published literature might suggest — and perhaps closer to resolution than is widely appreciated to date.
Toward a More Complete Picture
The Yamagata University study represents exactly the kind of rigorous, clinically grounded pharmacokinetic research that the field of PRRT needs. Its contribution is real: a simple, validated formula that can inform discharge planning and support the expansion of outpatient treatment in Japan and beyond.
And it points, implicitly, toward the next set of questions.
If tumor diameter and renal function predict surface dose rate decline, what predicts the variation that remains unexplained? If urinary excretion is the dominant elimination pathway, what does direct measurement of that excretion add to our understanding of individual kinetics? And if individual pharmacokinetic profiles vary as substantially as the literature suggests, what would it mean — for research, for monitoring, for the longer-term ambitions of precision medicine in nuclear oncology — to measure them more directly?
These are questions for researchers and clinicians to pursue. We find them worth asking.
Reference [1] Ono T, Ichikawa M, Tanada T, Kanezawa C, Sato H. Maximum tumor diameter and renal function can predict the declining surface dose rate after ¹⁷⁷Lu-Dotatate: preliminary results of single institution in Japan. Japanese Journal of Radiology. 2024;42:1031–1037. https://doi.org/10.1007/s11604-024-01585-5
This content is prepared for general academic discussion purposes only. It does not constitute medical advice, clinical guidance, or diagnostic recommendations. Treatment decisions must be made exclusively by licensed medical professionals. This content does not constitute promotion of any product or service. AMS Planning, Inc. makes no warranty regarding the completeness or accuracy of the information presented.
About the Author: Komei Washino, VMD, Ph.D., is Chief Technology Officer at AMS Planning, Inc. He brings 30 years of experience in the development, research, and commercialization of radiopharmaceuticals. Prior to joining AMS, he served as Director at Nihon Medi-Physics, held an appointment as Research Professor at Hokkaido University Graduate School of Medicine, and served as Senior Researcher at the National Institutes for Quantum and Radiological Science and Technology (QST). He received his VMD and Ph.D. from the Faculty of Veterinary Medicine, Hokkaido University.