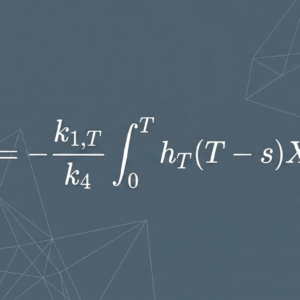Komei Washino, VMD, Ph.D. Chief Technology Officer, AMS Planning, Inc. Former Director, Nihon Medi-Physics | Research Professor, Hokkaido University Graduate School of Medicine | Senior Researcher, National Institutes for Quantum and Radiological Science and Technology (QST)
Historically, urine has been a basic barometer of health. Today, it also serves as a subject of significant interest in general pharmacokinetic research. Based on academic literature on radiopharmaceuticals and general pharmacokinetic concepts, here is a brief overview of how urinary excretion data is explored in current research settings.
Academic Observations: Urine and Pharmacokinetic Distribution
In certain targeted radionuclide therapies, patients receive Lutetium-177 (¹⁷⁷Lu)-based treatments, and a significant portion of the administered radioactivity is excreted in urine. Academic research in this area has begun to explore whether patterns in urinary excretion might reflect underlying differences in individual pharmacokinetic distribution.
Recent academic literature has observed notable variation in ¹⁷⁷Lu excretion rates across patients. Researchers have noted that patients with higher urinary excretion and patients with lower urinary excretion appear to show different patterns of residual radioactivity in the body. As all patients in the observed cohorts had comparable renal function, researchers have hypothesized that factors beyond renal clearance — such as differences in tissue distribution — may contribute to this variation.
This remains an area of active academic inquiry. Whether and how such observations might eventually inform research protocols is a subject for further investigation by qualified researchers and clinicians.
Urine Data in Pharmacokinetic Research
Studying compounds excreted in urine is an established approach in pharmacokinetic research across many therapeutic areas. Several dimensions of this research are worth noting from an academic perspective.
Pharmacokinetic profiling: Researchers study urinary excretion as one component of understanding how compounds distribute through the body. Excretion data contributes to broader pharmacokinetic models that describe absorption, distribution, metabolism, and elimination.
Individual variation: Academic literature increasingly documents inter-individual variability in pharmacokinetic parameters. Understanding the sources of this variability — genetic, physiological, disease-related — is a central question in pharmacokinetic research.
Precision medicine research: There is growing academic interest in whether individual pharmacokinetic profiles can inform research into personalized therapeutic approaches. Urinary excretion is one of several non-invasive data points being explored in this context.
These represent areas of academic and scientific inquiry. They do not represent established clinical protocols or validated diagnostic approaches.
Conclusion
Analyzing how and where a compound is excreted continues to be a subject of academic interest across therapeutic areas, including radiopharmaceuticals. Urinary excretion data represents one of several research variables being explored in the broader context of pharmacokinetic science.
Further research is needed to understand the significance and potential applications of these observations. The field remains at an early and exploratory stage.
Disclaimer: This content is prepared for informational and general academic discussion purposes only. It does not constitute medical advice, clinical guidance, or diagnostic recommendations. All medical evaluations and treatment decisions must be made exclusively by licensed medical professionals. AMS Planning, Inc. does not provide clinical judgment or imply any clinical effect based on the information presented herein. This content does not constitute a promotion or commercial advertisement of any product. The information contained herein was obtained from sources believed to be reliable; however, no representation or warranty is made that it is accurate or complete. AMS Planning, Inc. accepts no responsibility for the use of or reliance on the information provided.