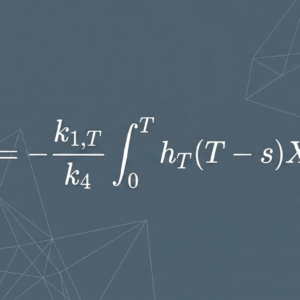After receiving ¹⁷⁷Lu-based therapy, patients excrete a significant portion of the administered radiopharmaceutical through their urine in the hours following treatment. In most clinical settings, this is managed as a radiation safety matter — and the urine is discarded. What is less widely discussed is the pharmacokinetic information that may be embedded in that excretion pattern, and what the research community is beginning to observe about its potential significance.
The Biology of Urinary Excretion in Radionuclide Therapy
When ¹⁷⁷Lu-labeled compounds are administered, the radiopharmaceutical distributes through the body according to its binding affinity and individual patient physiology. A portion binds to target tissue; the remainder undergoes renal clearance and is excreted in urine.
Academic research has begun to explore whether the proportion excreted in urine — and the variation observed across patients — may reflect differences in individual pharmacokinetic distribution. Recent literature has noted that patients show meaningful variation in urinary excretion rates following ¹⁷⁷Lu administration, even among those with comparable renal function. Researchers have hypothesized that this variation may reflect differences in how the compound distributes within the body beyond what renal function alone would explain.
These are early-stage academic observations. Their significance and implications remain subjects of ongoing inquiry.
An Overlooked Research Variable

The reason urinary excretion data has received limited systematic attention in nuclear medicine research is largely practical. Post-treatment patient urine is classified as radioactive waste, and the logistical and safety requirements involved in collecting and analyzing it have made routine study difficult in most settings.
The result is that a potentially informative research variable is routinely discarded — not because the research community has concluded it lacks interest, but because the infrastructure to study it systematically has been limited.
As the field of nuclear medicine grows — driven by the expanding use of ¹⁷⁷Lu-based therapies — academic interest in this variable is increasing. Researchers are asking whether systematic collection and analysis of post-treatment urinary excretion data could contribute to the pharmacokinetic knowledge base in ways that have not yet been fully explored.
What Pharmacokinetic Research Asks of Urine Data
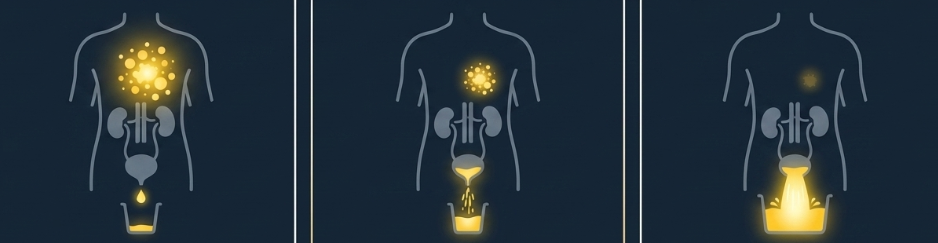
Urinary excretion analysis is an established method in pharmacokinetic research across many therapeutic areas. In the context of radionuclide therapy, researchers are exploring several questions.
How much individual variation exists in excretion patterns, and what factors drive it? What does variation in excretion tell us about how a compound distributes across different patients? How might systematic excretion data contribute to broader pharmacokinetic models being developed in the field?
These questions are at the research stage. They represent directions of academic inquiry rather than established clinical applications.
Conclusion
The growth of ¹⁷⁷Lu-based therapies is generating increasing volumes of post-treatment urinary data in nuclear medicine departments worldwide. Much of this is currently treated as waste.
The research community is beginning to ask whether this represents a missed opportunity — not as a clinical tool, but as a source of pharmacokinetic data that has been present all along, waiting for the means and the infrastructure to be studied systematically.
What we choose to measure, and what we choose to discard, shapes the questions we are able to ask. In nuclear medicine research, post-treatment urine may be one variable worth a closer look.
Disclaimer: This content is prepared for informational and general academic discussion purposes only. It does not constitute medical advice, clinical guidance, or diagnostic recommendations of any kind. All medical evaluations and treatment decisions must be made exclusively by licensed medical professionals. AMS Planning, Inc. does not provide clinical judgment or imply any clinical effect based on the information presented herein. This content does not constitute a promotion or commercial advertisement of any product or its efficacy. The information contained herein was obtained from sources believed to be reliable; however, no representation or warranty is made that it is accurate or complete. AMS Planning, Inc. accepts no responsibility for the use of or reliance on the information provided.